






Watch For Free are lewis structures only for covalent bonds pro-level online video. Without any fees on our video portal. Step into in a vast collection of series showcased in excellent clarity, excellent for prime watching mavens. With recent uploads, you’ll always keep current. Seek out are lewis structures only for covalent bonds curated streaming in vibrant resolution for a truly enthralling experience. Become a patron of our creator circle today to stream restricted superior videos with without any fees, no strings attached. Get access to new content all the time and uncover a galaxy of indie creator works created for premium media connoisseurs. Be certain to experience never-before-seen footage—begin instant download! Explore the pinnacle of are lewis structures only for covalent bonds special maker videos with exquisite resolution and curated lists.
Lewis structures for polyatomic ions follow the same rules as those for other covalent compounds. Lewis structures we also use lewis symbols to indicate the formation of covalent bonds, which are shown in lewis structures, drawings that describe the bonding in molecules and polyatomic ions For example, when two chlorine atoms form a chlorine molecule, they share one pair of electrons The lewis structure indicates that each cl atom has three pairs of electrons that are not used in bonding.
30 7.3 lewis structures and covalent compounds learning objectives by the end of this section, you will be able to Illustrate covalent bond formation with lewis electron dot diagrams Draw lewis structures depicting the bonding in simple molecules Lewis structures formation of covalent bonds can be represented using lewis symbols
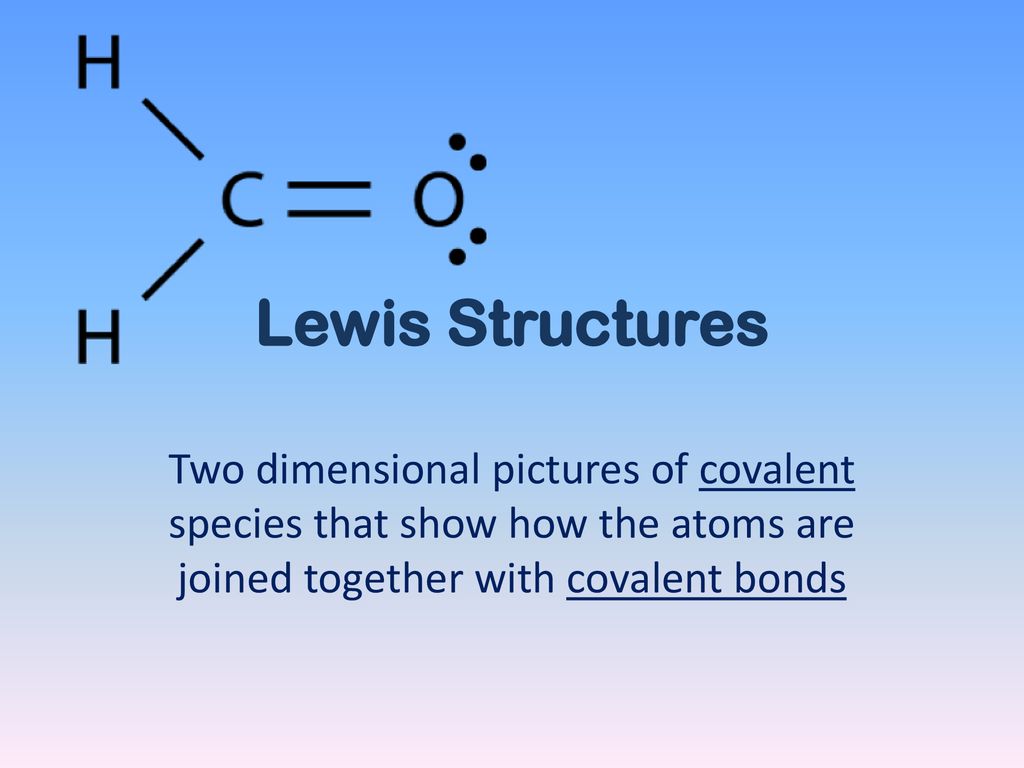
We usually show each electron pair shared between atoms as a line and show unshared electron pairs as dots Each pair of shared electrons constitutes one chemical bond. What is lewis structure lewis structure, also known as lewis dot structure or electron dot structure, is a simple and straightforward way of representing the outermost electron shell in a chemical species like an atom, ion, or molecule It shows how electrons are positioned around the atoms either as lone pairs or in a chemical bond, typically a covalent bond or a coordinate covalent bond.
The lewis structures illustrated so far have been selected for their simplicity A number of elaborations are given below There is sometimes an ambiguity in the location of double bonds This ambiguity is illustrated by the lewis structure for ozone (o3)
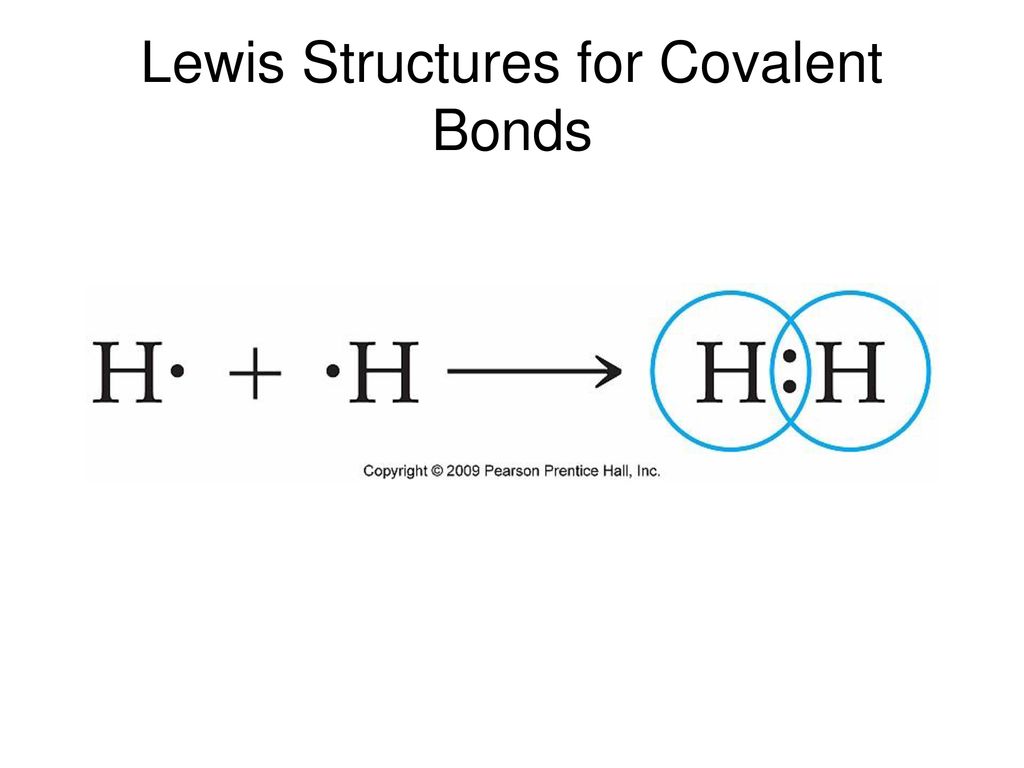
In such cases, the actual lewis. Lewis proposed that atoms combine in order to achieve a more stable electron configuration Maximum stability results when an atom is isoelectronic with a noble gas An electron pair that is shared between two atoms constitutes a covalent bond.

The Ultimate Conclusion for 2026 Content Seekers: To conclude, if you are looking for the most comprehensive way to stream the official are lewis structures only for covalent bonds media featuring the most sought-after creator content in the digital market today, our 2026 platform is your best choice. Seize the moment and explore our vast digital library immediately to find are lewis structures only for covalent bonds on the most trusted 2026 streaming platform available online today. We are constantly updating our database, so make sure to check back daily for the latest premium media and exclusive artist submissions. Enjoy your stay and happy viewing!
OPEN